How to perform an asymptomatic sexual health screen in primary care: our recommendations
Our most common question received at local and regional testing events is how to perform an effective and appropriate sexual health screen for asymptomatic patients requesting testing. Clearly one option is for those patients to attend our services for review, however we would encourage delivery of asymptomatic screening in other settings – primarily for the convenience of patients.
The information below will help support and inform testing at your service. We are happy to discuss and support any services with testing issues or concerns, as would your local microbiology laboratory.
Please also note that people in England under 25 years old can also get tested as part of the National Chlamydia Screening Programme (NCSP).

Chlamydia
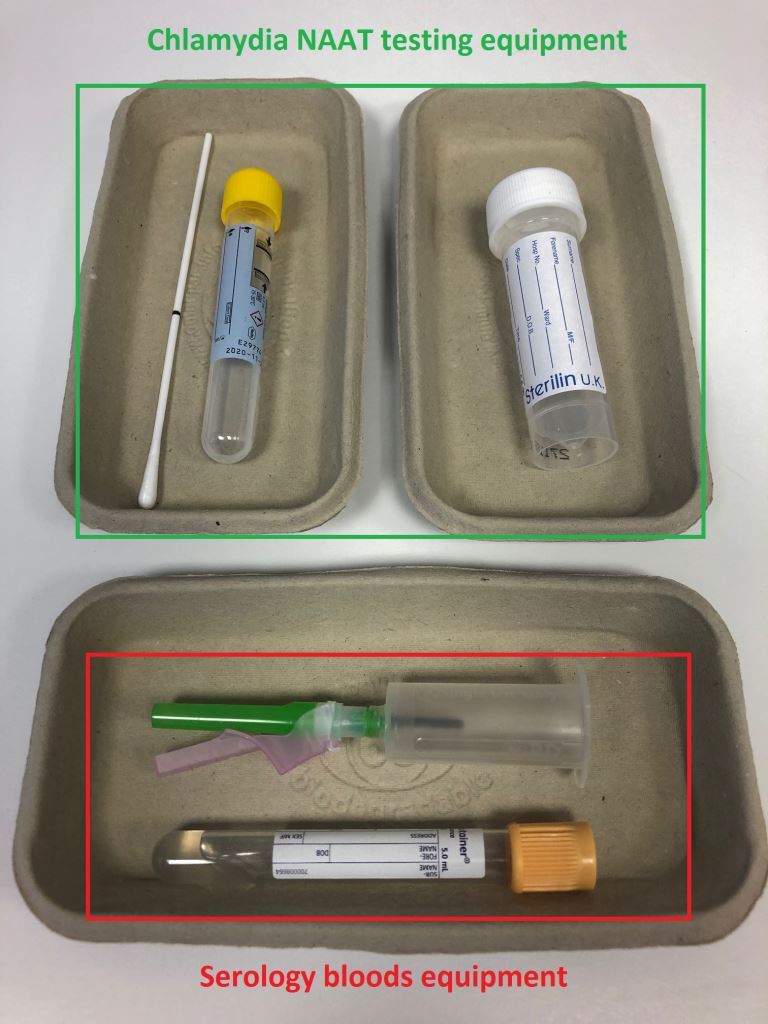
Nucleic acid amplification tests (NAATs) remain the most sensitive and specific test for chlamydia. Testing can be performed on any potential infected anatomical site but routine assessment in primary care requires genital testing only. Depending on the patient’s anatomy the sample to test does vary:
- Vagina – low vaginal swab – a NAAT testing swab applied liberally in and around the vaginal intraiotus for 10 seconds. This sample type will collect cervical discharge and residual urine – the two potential genital sites of infection. This sample can be self-taken.
- Penis– urine sample – a sterile urine pot (without boric acid) should be used to collect a first catch urine sample. The urine should have been held for at least 60, but ideally 90, minutes to maximize NAAT sensitivity. Urethral swabbing for NAATs is not preferred as it confers no improved sensitivity and is more uncomfortable for the patient.
Testing for trans patients
Testing should be adapted according to the anatomy of the patient.
Patients not currently seeking or awaiting reassignment surgery should be tested as above.
Following gender reassignment surgery, and the formation of a neovagina or neopenis, the sample of choice is a first catch urine as the only potential anatomical site for chlamydial infection is the urethra.

Gonorrhoea
Assessment for, and the management of, gonorrhoea is currently recommended to take place in sexual health services in England. This is due to increasing concern over antimicrobial resistance amongst circulating gonorrhoea strains internationally, and also due to the increased chance of false positive results when compared with chlamydia screening.
Currently if you wish to test for gonorrhoea in primary care this can be attempted via direct culture from the affected site[s]: ie. cervix and/or urethra. Specific gonococcal culture should be requested and the sample transported to the lab immediately as Neisseria gonorrhoea is a particularly friable organism.
UPDATE: Following an ongoing and sustained outbreak of gonorrhoea in early 2023 gonorrhoea NAAT testing will now be available for samples sent from primary and secondary care.

Blood testing
NICE guidance recommends that a sexual health screen in England should also include the offer of serological testing for HIV and syphilis.
A common misconception amongst clinicians and patients is that detailed verbal consent is required to perform an HIV test. Whilst this was the case in the 1990s it is no longer the case and is considered a significant barrier to offering testing. Verbal consent is still required but is no more detailed than asking for permission to perform the test, explaining it is a blood test, and how results will be communicated to the patient once they are available.
Further assessment for blood borne virus transmission occurs as part of routine reviews in sexual health services. This can inform a decision in some patients to test for Hepatitis A, B and/or C. If time permits during primary care attendances discussions around common risk factors such as intravenous drug use and endemic sexual contacts.

Window periods
A window period is the time from acquiring infection until a timepoint where its screening test has the highest sensitivity.
- For chlamydia infection this time is 2 weeks.
- For HIV and syphilis serology this window is currently 4-6 weeks.
Serological testing currently has a 99% sensitivity at 6 weeks, and close to 100% sensitivity at 3 months.
Testing equipment